Electronic energy levels in simple crystalline solids have a bandstructure to them. (Bandstructure is just energy vs. wavevector or momentum.) Depending on the filling of the bands, the material can either become a metal, insulator, or semiconductor. Metals have partially filled bands. Insulators and semiconductors have a filled band at zero temperature, with an energy gap to the next band. Good insulators have such a large gap (about 5eV or more) that even room temperature is not enough to excite electrons across the gap into the next highest energy band. But semiconductors have lower band gaps (about 1eV), so that at room temperature, there are many electrons excited into the next band. The missing electrons in the lower band are called holes. Holes aren't real particles, they're just missing electrons -- but we can treat them as if they were real particles with positive charge. We also introduce how to dope semiconductors into n-type and p-type semiconductors. Lecture Audio
Show More
Unlock more with Podchaser Pro
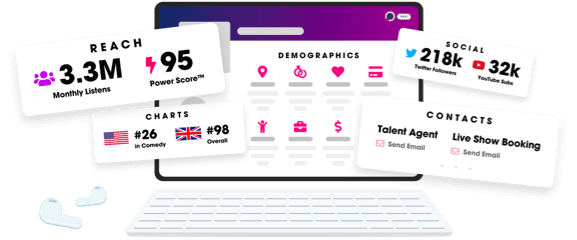
- Audience Insights
- Contact Information
- Demographics
- Charts
- Sponsor History
- and More!
- Account
- Register
- Log In
- Find Friends
- Resources
- Help Center
- Blog
- API
Podchaser is the ultimate destination for podcast data, search, and discovery. Learn More
- © 2025 Podchaser, Inc.
- Privacy Policy
- Terms of Service
- Contact Us
